Privacy Concerns Arise over Storage of Millions of Newborns’ Blood Samples Used for Mandatory U.S. Disease Screenings
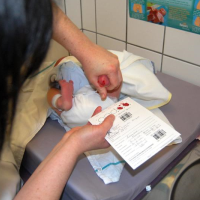 Blood sample drawn from newborn's heel (photo: Wikimedia Commons)
Blood sample drawn from newborn's heel (photo: Wikimedia Commons)
By Rick Callahan, Associated Press
INDIANAPOLIS (AP) — Two-day-old Ellie Bailey squirms in a hospital bassinet and cries as her tiny left heel is squeezed and then pricked with a needle to draw a blood sample. An Indianapolis hospital technician quickly saturates six circles on a special filter card with the child's blood.
Ellie is among some 4 million newborns in the United States who will have blood drawn this year to screen them for serious inherited diseases such as sickle cell anemia, which can cause organ damage, and the metabolic disease phenylketonuria, or PKU, which can lead to mental disabilities.
Newborn screening saves or improves the lives of about 12,000 newborns each year by swiftly identifying therapies they might need, said Jelili Ojodu, director of newborn screening and genetics with the Association of Public Health Laboratories.
But what happens to the dried blood samples on those cards after the testing that's mandatory in all 50 states is completed has sparked legal battles in some states. Minnesota and Texas have destroyed some 6.4 million samples following lawsuits. And in Indiana, the parents of a 9-year-old suburban Indianapolis girl are seeking the same for up to 2.5 million samples collected over two decades and stored in 600 boxes at a state warehouse.
"Her parents' main concern is that down the road who knows what could happen with these samples?" said Jonathan Little, an attorney for parents of the girl, identified in court documents only as A.B. Doe.
The case poses a dilemma: How can society balance the right to privacy with the needs of science and medical research?
The Indiana lawsuit was dismissed by a trial court last year and in April by the Indiana Court of Appeals. Both courts found the girl was in no imminent danger of suffering harm from the state holding onto her sample. Her attorneys plan to appeal to the Indiana Supreme Court next week.
In an era of increasingly sophisticated genetic analysis, some privacy advocates fear insurance companies could access blood samples and charge higher premiums for people found to have a genetic predisposition to diseases such as Alzheimer's.
"The question is, 'These cards are being collected for a very specific purpose, and once they have met that purpose what's the necessity for keeping these blood stains?" said Sheldon Krimsky, acting executive director of the Cambridge-Massachusetts-based Council for Responsible Genetics.
He said law enforcement could also potentially access states' newborn blood stores to use them to create DNA databases of law-abiding citizens.
Such worries are "really a far stretch" and not based on actual threats to a person's genetic privacy, said Natasha Bonhomme, vice president of strategic development for the Genetic Alliance, a Washington, D.C.-based nonprofit health advocacy group.
Scott Kelly, an attorney who represented some of the families who sued Minnesota over its blood-retention policies, said more than 50,000 of Minnesota's dried blood samples and test results were used for research, including third-party research by drug companies and manufacturers. That suit led Minnesota in 2014 to destroy 1.1 million newborn blood samples and another 900,000 test results.
In 2009, Texas destroyed some 5.3 million newborn blood samples as part of a settlement of a similar lawsuit, and Texas lawmakers also set into law an opt-out policy for parents who object to the state using their child's blood for research.
Attorneys for Indiana say the 2.5 million blood samples at the heart of the pending lawsuit won't be used for medical research, but argue the state has an interest in holding onto them, such as for evidence in missing persons or medical malpractice cases.
Starting in June 2013, Indiana tried to solve the dilemma by asking the parents of newborns for permission to use their infant's blood for medical research. The parents of just over 63,000 infants — about a quarter of the babies born in Indiana in the past three years — have consented to that, said Jennifer O'Malley, an Indiana State Department of Health spokeswoman.
Those now include 27-year-old Jessica Bailey — the mother of Ellie. She checked the "yes" box on Indiana's consent form and signed it Thursday, authorizing the state to use her daughter's blood for medical research.
She and her husband, Mark, said they don't share concerns about the blood's possible misuse and believe there's great potential in allowing its use for medical research.
"I personally feel the benefits with the research outweigh the risks," Jessica Bailey said as she and Mark prepared to leave Community Hospital North in Indianapolis with their second child. "For us, It was kind of an easy decision because it might help somebody in the future."
Indiana has posted a form on the state health department's website allowing parents to request that their child's blood sample be destroyed, no matter when it was collected. That's not enough for the father of the 9-year-old girl who sued the state. He asked The Associated Press not to use his name because it would identify his daughter.
He said he supports disease-screening for newborns, and said his daughter has even taken part in a medical research project for stomach ailments. But he believes it's time for the state to dispose of the blood samples collected between 1991 and May 2013 that it held onto without alerting parents.
"Somebody obviously has some kind of plans for it, if they've gone to the expense of storing it and keeping it," he said.
To Learn More:
DNA Privacy Bill Targets Massive State Biobank Holding 16 Million Samples (by Ken Broder, AllGov California)
- Top Stories
- Unusual News
- Where is the Money Going?
- Controversies
- U.S. and the World
- Appointments and Resignations
- Latest News
- Trump Kidnaps Gov. Newsom and His Wife
- Trump Orders ICE and Border Patrol to Kill More Protestors
- Trump Renames National Football League National Trump League
- Trump to Stop Deportations If…
- Trump Denounces World Series






Comments